  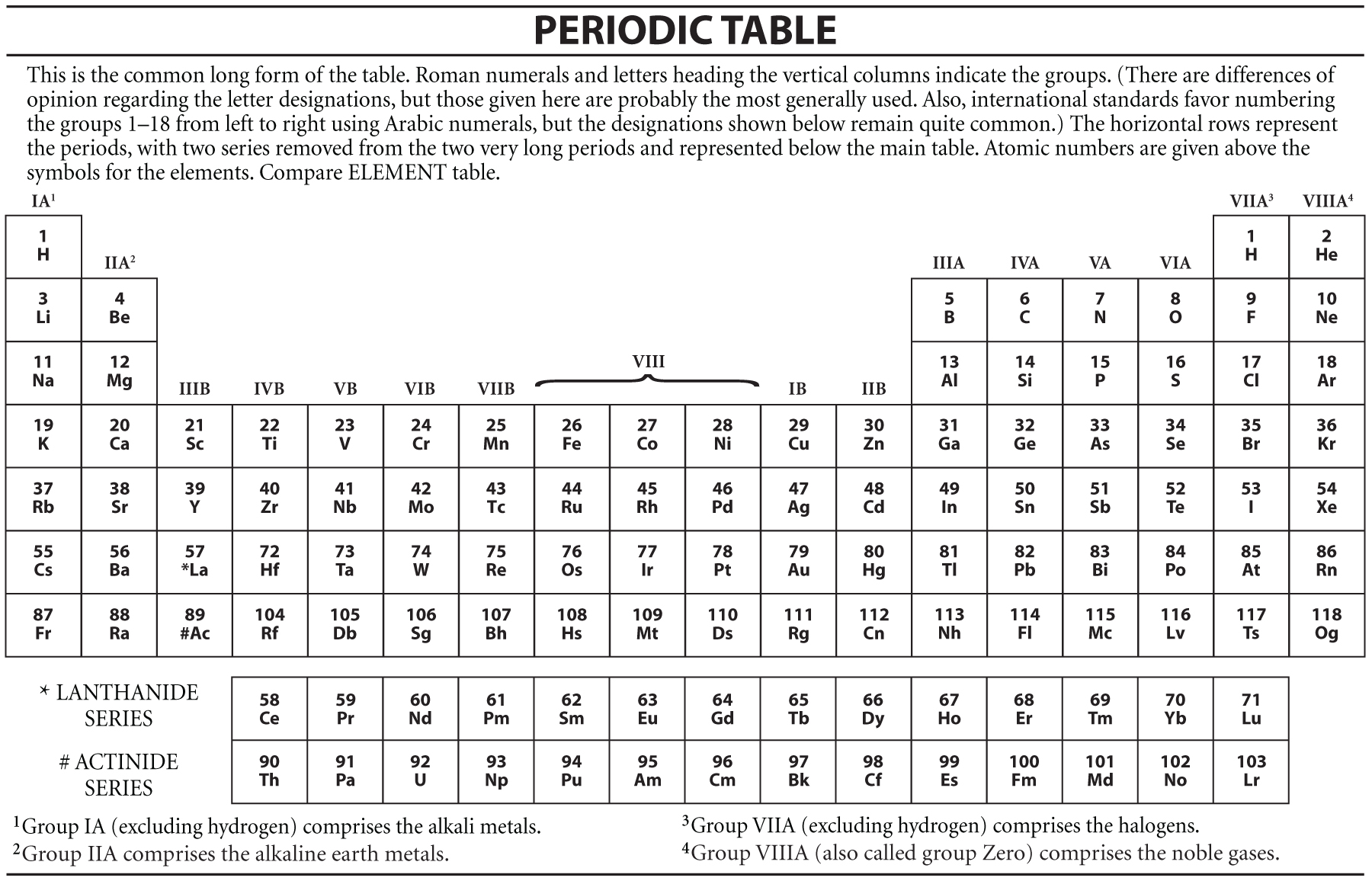
Transition metals are used as building materials and in electronics. Transition metals also form ions with multiple oxidation states, form complex ions, produce coloured compounds, and act as catalysts. Transition metals are hard and strong, have high melting and boiling points, and are relatively unreactive. When forming ions, transition metals lose their 4s electrons before their 3d electrons. However, copper and chromium have slightly different electron configurations than expected due to the similarity in energy levels of the 4s- and 3d-subshells. Transition metals differ by the number of electrons in their d-subshells. More specifically, transition metals are found in groups 3 -12 and periods 4-7. This means that their highest energy subshell is always a d-subshell. Transition metals are found in the d-block of the periodic table. Transition metals are elements whose atoms have a partially filled d-subshell, or which form at least one stable ion with a partially filled d-subshell of electrons. 5 - The electron configuration of iron, iron(II) and iron(III) Since the 4s-subshell is now empty, this electron is lost from the 3d-subshell, giving the ion the electron configuration of 3d 5 4s 0.įig. 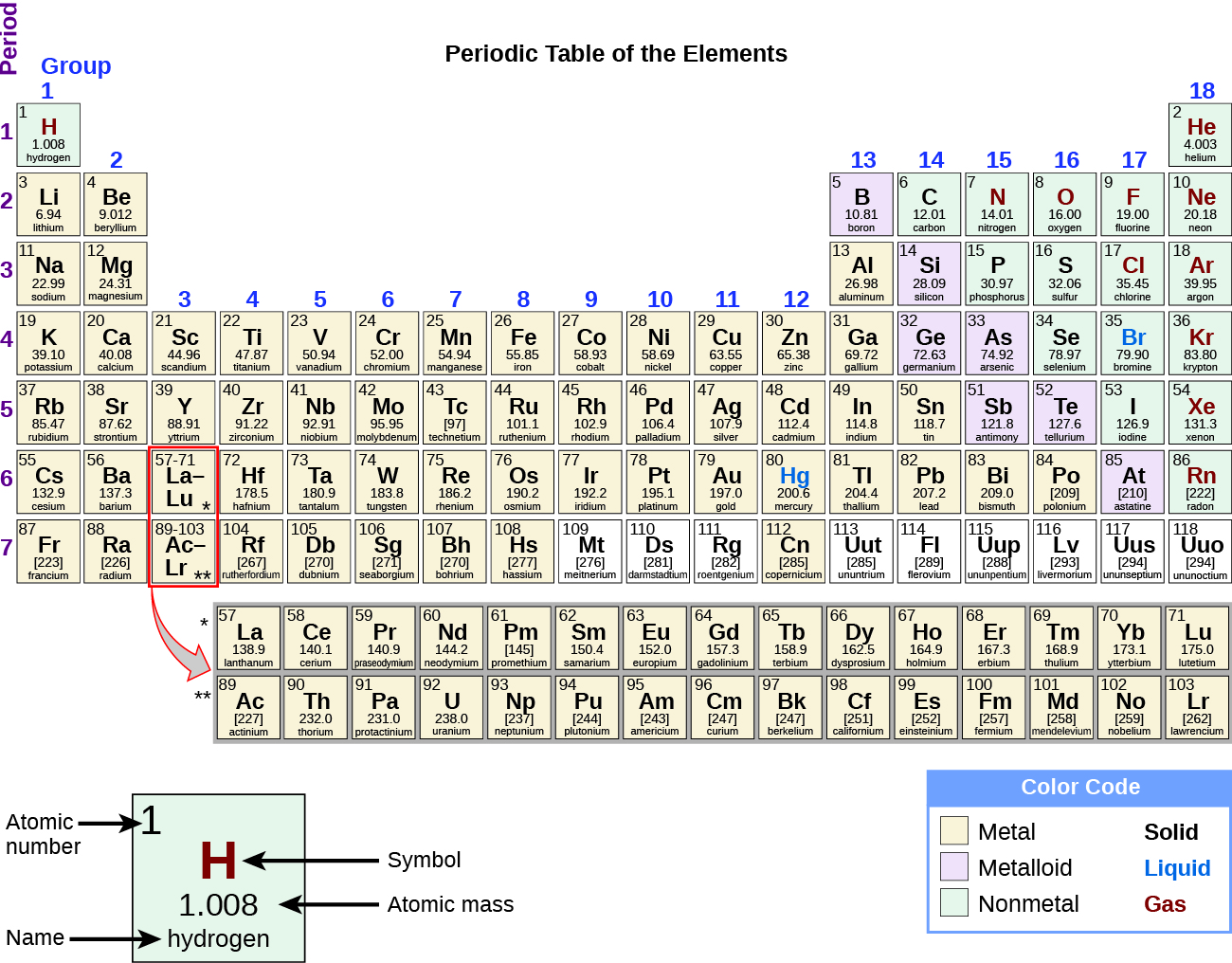
To form a 3+ ion, it needs to lose a further electron. When forming a 2+ ion, it first loses its 4s electrons, giving it the electron configuration of 3d 6 4s 0. Iron has the electron configuration of 3d 6 4s 2. It commonly forms ions with charges of 2+ or 3+. This means that all transition metals lost their 4s electrons before their 3d electrons. You might remember from Electron Configuration that although the 3d-subshell is of a slightly higher energy level than the 4s-subshell, atoms lose electrons from the 4s-subshell first. 4 - Expected and observed electron configurations of chromium and copper Electron configuration of transition metal ionsĪll transition metals form positive cations by losing electrons. It is also believed that having a half-full 3d-subshell, as in the case of chromium, or a completely filled 3d-subshell, in the case of copper, helps stabilise the atom.įig. Electrons simply like being in the lowest energy state possible. This lowers its energy state and more than makes up for the extra electron in the slightly higher energy 3d-subshell. Since the electron in the 4s-subshell is unpaired, it doesn't experience any electron-electron repulsion. Well, it is because the 4s- and 3d-subshells have very similar energy levels. This gives it the electron configuration of 3d 2 4s 2.īut as we mentioned above, this filling pattern is rudely interrupted by two elements: chromium (Cr) and copper (Cu). For example, scandium (Sc) has 21 electrons and has just one electron in its 3d-subshell, giving it the electron configuration of 3d 1 4s 2, whereas titanium has 22 electrons and has two electrons in its 3d-subshell. As you go across the period, electrons are added to the inner 3d-subshell, one by one. The next 10 elements are d-block elements. This is just another example of an annoying exception to the rules that you need to learn! However, 3d is an anomaly - it has a slightly higher energy than 4s and so fills up after 4s. This usually follows the pattern of lowest number to highest number. Remember that subshells fill up in a certain order, from lowest energy to highest energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
